One of the luxuries that I am most grateful for is the opportunity to be a full-time, stay-at-home mum. Some might think it’s decadent, given that my youngest child is already 16 and her older siblings (eldest one is 31) all live abroad. But apart from my time at home with my other children and my parents, the rest of my time is wholly dedicated to Georgina, as is her father’s. She is our last offspring to cap off our long parenting journey and we are savouring every moment of our time with her. We both would be in the car together as much as possible when we ferry her round, be it to football practice or her boyfriend’s house or even just to a party down the road. During the car journeys, we talk non-stop about many things. I think her “real” education happens here. Below is an example.
******
So, what do you remember about Chemistry from your own school days? Many of you would say remembering chemical reactions (what colour when you mix A with B, etc). Many of you would say equations. Many of you would say test tubes.
I had a wonderful teacher called Cliff Haskins, an Oxford man. He would tell us, “Just remember the first 15 minutes. We can talk about other things after that.” Little did I know then, but dear Mr Haskins actually worked very hard before each class to put all we need to know for a particular topic into 15 minutes of teaching time. Because we had such a sweet deal with him, we always paid rapt attention for the first 15 minutes. The other 45 minutes, well, we spent talking. Either gossiping with each other (he never minded) or taking part in his interesting, offbeat discussions.
I decided to teach my child this way. She had to learn benzene in class today. but here is what I was preparing at home for us to have fun with.
Step 1: Getting excited about C6H6
What’s so special about benzene? Its structure, of course. Try drawing C6H6, taking into consideration the covalence of C and H. What did you get? Scientists couldn’t figure out what it looked like for a long time. Codswallop about dreams of snakes swallowing each other’s tails and 6 monkeys holding hands. Finally, it was proved by looking at the bond lengths and Delta H.
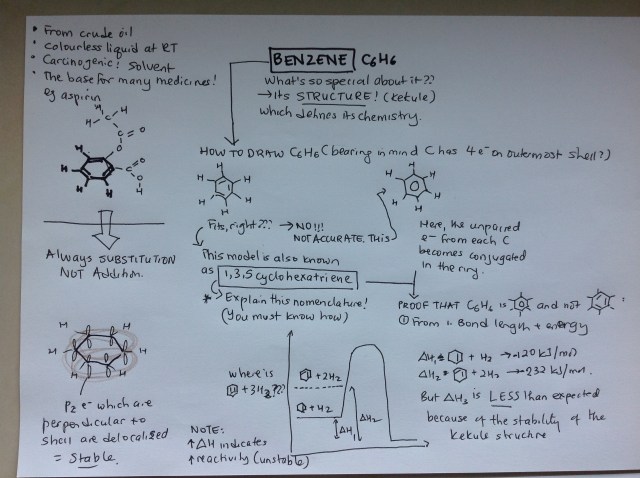
Step 2: So what?
Its shape gives it its special properties. It does not undergo addition, but substitution happens quite a lot. Aspirin, paracetomol, solvents. They are all benzene-based. Sorry dear child, you have to memorise the key reactions, but I have summarised the key points for you. It’s not too bad if you print this out and stick it on your wall. Look at it before you fall asleep at night instead of your boyfriend’s photograph.
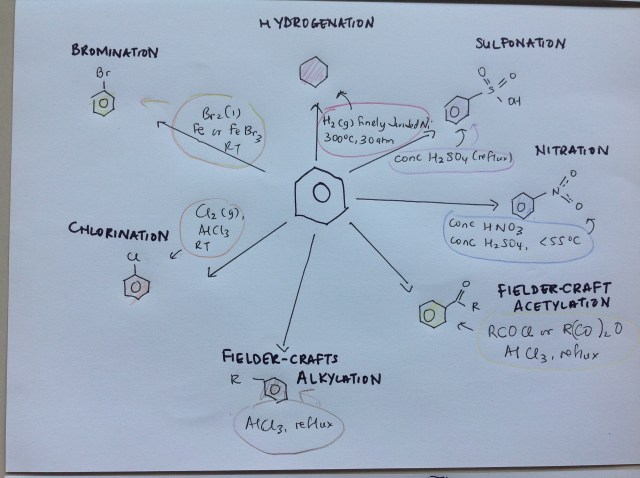
Step 3: Let the fun begin!
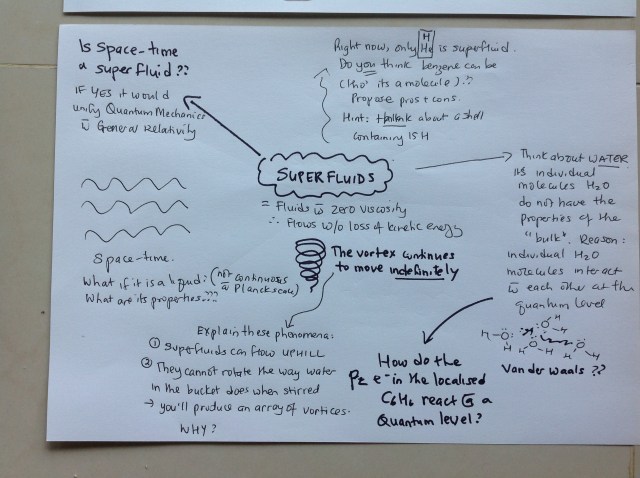
In my book, Catching Infinity, I wrote that exciting things happen at the boundaries. That’s why daredevils leap off tall buildings and biplanes. But we can do the same sitting in the comfort and safety of our homes IF we allow our brains to leap into the unknown. Real education after all is about exploring and thinking the improbable, rather than memorising. So I put this to my child: think about the extraordinary properties of benzene because of its delocalised pz electron cloud. Now think about superfluids. Can benzene possibly be a candidate for superfluids? And imagine what a world with a benzene-like spacetime feel like? Would it be like Alice In Wonderland’s treacle world?
Yes, I was wrong to criticise the International Baccalaureate syllabus. After all, she goes to school to learn the rudiments to pass exams. It is up to me, her parent, to teach her about excitement and the boundless possibilities, and I am loving the journey.